Developing Emission Reduction Strategies
8.2 On-Road Mobile Sources
8.2.3 Emissions Control Technology for Diesel Vehicles
Diesels emit high levels of oxides of nitrogen and particulates as noted earlier. Modest to significant NOx control can be achieved by delaying fuel injection timing and adding exhaust gas recirculation (EGR). Very high pressure, computer controlled fuel injection can also be timed to reduce PM emissions. (Modifying engine parameters to simultaneously reduce both NOx and PM is difficult and limited since the optimal settings for one pollutant frequently increases emissions of the other and vice-versa.) Very low levels of NOx and PM therefore require exhaust treatment. Lean NOx catalysts, selective catalytic reduction, NOx storage traps with periodic reduction, filter traps with periodic burn-off, and oxidation catalysts with continuous burn-off are evolving technologies that are being phased in at differing rates in various parts of the world. Japan for example, is tending to lead the world in the widespread use of PM filters on new diesel vehicles whereas Europe is tending to lag. Some European countries are using tax incentives to accelerate the introduction of PM filters beyond the rate required by the Euro new vehicle standards.
A new type of diesel, the homogeneous charge compression ignition engine, provides another approach to reducing NOx and particulates that is receiving significant attention and may be introduced on some engines for at least portions of the engine map within a few years.
Reformulated diesel fuels can effectively reduce oxides of nitrogen and particulate emissions from all diesel vehicles. These fuels have reduced sulfur, reduced aromatics, and increased Cetane Number. However, certain technologies are especially sensitive to the sulfur content of the fuel; the linkages between sulfur and diesel vehicles technologies will be summarized below.
8.2.3.1 No Controls/Pre-Aftertreatment Controls
For diesel vehicles with no controls, the amount of sulfur in the fuel is directly related to SO2 and PM emissions; some SO2 emissions are converted in the atmosphere to sulfate PM.
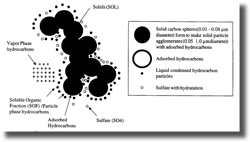
The amount of SO2 emissions is directly proportional to the amount of sulfur contained in the fuel. In addition, total PM emissions are proportional to the amount of sulfur in the diesel fuel although the carbon and the soluble organic fractions are not affected. In the oxygen-rich exhaust of diesel vehicles several percent of the SO2 formed during combustion is oxidized to SO3, which dissolves in the water vapor present to form sulfuric acid (H2SO4) vapor. H2SO4 forms very small (so called ultrafine) particles in diesel exhaust which are considered especially hazardous because of their ability to penetrate deeply into the lungs. Even though sulfate particles account for only a small fraction of particle volume or mass, they account for a large fraction of particle numbers.
According to the US EPA, approximately 2% of the sulfur in the diesel fuel is converted to direct PM emissions. In addition, SO2 emissions can lead to secondary particle formation—particles that form in the ambient air. US EPA models predict that over 12% of the SO2 emitted in urban areas is converted in the atmosphere to sulfate PM. Urban areas would benefit most from reductions in SO2 emissions, as polluted urban air has higher concentrations of the constituents that catalyze the SO2-to-sulfate reaction. Even with vehicle stocks without advanced pollution controls, reductions of fuel sulfur levels would likely have a significant impact on primary and secondary PM concentrations in urban areas.
8.2.3.2 Post Combustion Controls
With high sulfur levels, diesel catalysts produce high levels of hazardous sulfate. Some advanced catalyst technologies such as NOx adsorbers are precluded by high levels of sulfur. Finally, PM filter performance is impaired by higher levels of sulfur.
8.2.3.3 Diesel Oxidation Catalysts
Diesel oxidation catalysts (DOCs) are the most common aftertreatment emissions control technology found in current diesel vehicles. DOCs are very similar to the earliest catalysts used for gasoline engines. Oxidation catalysts work by oxidizing CO, HC and the soluble organic fraction of the PM to CO2 and H2O in the oxygen rich exhaust stream of the diesel engine.
When sulfur is present in the fuel, DOCs also increase the oxidation rate of SO2, leading to increases in sulfate nanoparticle emissions. Sulfate conversion depends on overall catalyst efficiency, with more efficient catalysts capable of converting nearly 100% of the SO2 in the exhaust to sulfate. Generally, one should restrict the use of DOCs to areas which have fuel sulfur levels of 500 PPM or below. With low sulfur fuel, a DOC can reduce PM emissions by 25 to 30%.
8.2.3.4 Diesel Particulate Filters
Diesel particulate filters (DPFs) already reliably demonstrate over 95% efficiency with near-zero sulfur fuel use. They are also capable of reducing the total number of particles emitted to levels similar or even slightly lower than those of gasoline engines. One important area of research—the area most impacted by sulfur levels—is the passive regeneration or cleaning of the collected particles from the filter surface. Filters need to be cleaned, ideally without human intervention, before reaching capacity in order to maintain vehicle performance and fuel and filter efficiency.
The Continuously Regenerating Diesel Particulate Filter (CR-DPF) and the Catalyzed Diesel Particulate Filter (CDPF) are two examples of PM control with passive regeneration. The CR-DPF and CDPF devices were found to achieve 95% efficiency for control of PM emissions with 3 ppm sulfur fuel (see DECSE study). But efficiency dropped to zero with 150 ppm sulfur fuel and PM emissions more than double over the baseline with 350 ppm sulfur fuel. The increase in PM mass comes mostly from water bound to sulfuric acid. Soot emissions also increase with higher sulfur fuel but even with the 350 ppm sulfur fuel DPFs maintain around 50% efficiency for non-sulfate PM. The systems eventually recover to original PM control efficiency with return to use of near-zero sulfur fuels, but recovery takes time due to sulfate storage on the catalyst.
As noted by TERI in its recent study of PM filters and low sulfur fuel in Mumbai, “CRT technology proved to be highly effective in reducing PM emissions from ULSD powered BS -II buses. It is, however, important to highlight that CRT is very sensitive to the sulphur content in diesel. According to JM, its manufacturer, a CRT can work effectively only if it is used in a modern diesel bus running on not more than 50 ppm sulphur–diesel or ULSD. The conversion efficiency of the CRT, after it was stabilized, was found to be 95% for SOF and over 98% for IOF . . . The CRT was very effective in reducing the free acceleration smoke too” (“Workstream 1,” 2004).
Sulfur also increases the required temperature for regeneration of the filter. In moving from 3 to 30 ppm sulfur fuel, the exhaust temperatures required for regeneration increase by roughly 25°C. The CDPF requires consistently higher temperatures but holds stable above 30 ppm, while the CR-DPF requires ever-increasing temperatures.
Work continues to develop filters that are less sensitive to sulfur in fuels. One emerging technology, the so-called flow through filter that achieves about 50% PM reduction is, for example, less sensitive to sulfur than the wall flow filter which can achieve 90% or greater PM reductions.
8.2.3.5 NOx Control Systems
Many diesel engines rely on injection timing retard to meet the NOx standards currently in place. Injection timing retard reduces the peak temperature and pressure of combustion, thus reducing NOx formation. Unfortunately, this solution both increases PM emissions and significantly decreases fuel economy. For example, NOx emissions can be decreased by 45% by retarding the injection timing 8 degrees, but this results in a 7% loss in fuel economy. Injection timing retard is not impacted by sulfur in fuel.
Exhaust gas recirculation (EGR), another NOx control strategy which is being used extensively today, is only indirectly impacted by fuel sulfur. Two very different technologies—NOx adsorbers and selective catalytic reduction (SCR) systems—are the most likely alternatives for stringent NOx control.
8.2.3.6 Exhaust Gas Recirculation
Major advances in diesel NOx control have been made with exhaust gas recirculation (EGR), which lowers combustion temperatures and thus reduces thermal NOx formation. Fuel sulfur does not impact emissions from EGR systems in diesel engines, but it does hinder system durability and reliability due to sulfuric acid formation. In order for EGR to be effective, the exhaust gases must be cooled, which causes sulfuric acid to condense in the recirculation system. Acid formation raises system costs, due to the need for premium components and increased maintenance costs.
8.2.3.7 Selective Catalytic Reduction (SCR)
SCR is emerging as the leading NOx reduction technology in Europe to meet Euro IV and Euro V heavy-duty diesel standards. SCR uses a reducing agent, injected into the exhaust gas before the catalyst, to achieve high rates of NOx conversion in the oxygen-rich exhaust. SCR systems are completely ineffective if the urea reagent is not added and thus requires great attention to in use enforcement and monitoring when this technology is used. European regulators are taking steps to require fail-safe systems that will significantly degrade vehicle performance if the urea tank is not filled. Stationary systems have over 90% conversion efficiency and are widely used for diesel generators and power production.
Sulfur does not reduce conversion efficiency in SCR systems as directly as in other advanced control technologies, but emissions are impacted in a couple of ways. Fuel sulfur will increase the PM emissions from the downstream oxidation catalyst. Sulfur reactions in urea-based SCR systems can also form ammonium bi-sulfate, a severe respiratory irritant.
8.2.3.8 NOx Adsorbers
NOx adsorbers are also known as NOx storage catalysts or lean NOx traps. NOx adsorber systems are still under development but are expected to be introduced in the US for some engines in 2007. They have demonstrated 95% efficiency in conversion of NOx to N2, with a nominal fuel penalty of 1.5%. However, without significant technological breakthroughs, it is generally recognized that this system can only operate with near zero sulfur fuels.
8.2.3.9 PM Retrofits
A growing body of data continues to show that the combination of very low sulfur fuel (usually with 50 ppm sulfur or less) and particulate filters can bring about approximately 90% reductions in PM and further substantial reductions in CO and HC from existing diesel vehicles, even after 400,000 miles of operation (“The Success of Diesel Retrofits: A Fuel Supplier Perspective”, Bob Schaefer, BP Global Fuels Technology, December 2003). To obtain these reductions however requires a careful matching of the technology to the vehicles with special attention given to operational patterns and exhaust temperature profiles.
Diesel oxidation catalysts can also be retrofitted to existing diesel vehicles as is occurring in Hong Kong, with overall PM reduction on the order of 25%.
A new type of diesel, the homogeneous charge compression ignition engine, provides another approach to reducing NOx and particulates that is receiving significant attention and may be introduced on some engines for at least portions of the engine map within a few years.
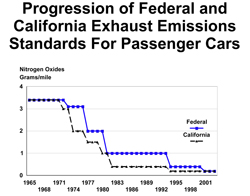
8.2.3-1 Progression of Federal and California Exhaust Emissions Standards for Passenger Cars
Reformulated diesel fuels can effectively reduce oxides of nitrogen and particulate emissions from all diesel vehicles. These fuels have reduced sulfur, reduced aromatics, and increased Cetane Number. However, certain technologies are especially sensitive to the sulfur content of the fuel; the linkages between sulfur and diesel vehicles technologies will be summarized below.
8.2.3.1 No Controls/Pre-Aftertreatment Controls
For diesel vehicles with no controls, the amount of sulfur in the fuel is directly related to SO2 and PM emissions; some SO2 emissions are converted in the atmosphere to sulfate PM.
The amount of SO2 emissions is directly proportional to the amount of sulfur contained in the fuel. In addition, total PM emissions are proportional to the amount of sulfur in the diesel fuel although the carbon and the soluble organic fractions are not affected. In the oxygen-rich exhaust of diesel vehicles several percent of the SO2 formed during combustion is oxidized to SO3, which dissolves in the water vapor present to form sulfuric acid (H2SO4) vapor. H2SO4 forms very small (so called ultrafine) particles in diesel exhaust which are considered especially hazardous because of their ability to penetrate deeply into the lungs. Even though sulfate particles account for only a small fraction of particle volume or mass, they account for a large fraction of particle numbers.
According to the US EPA, approximately 2% of the sulfur in the diesel fuel is converted to direct PM emissions. In addition, SO2 emissions can lead to secondary particle formation—particles that form in the ambient air. US EPA models predict that over 12% of the SO2 emitted in urban areas is converted in the atmosphere to sulfate PM. Urban areas would benefit most from reductions in SO2 emissions, as polluted urban air has higher concentrations of the constituents that catalyze the SO2-to-sulfate reaction. Even with vehicle stocks without advanced pollution controls, reductions of fuel sulfur levels would likely have a significant impact on primary and secondary PM concentrations in urban areas.
8.2.3.2 Post Combustion Controls
With high sulfur levels, diesel catalysts produce high levels of hazardous sulfate. Some advanced catalyst technologies such as NOx adsorbers are precluded by high levels of sulfur. Finally, PM filter performance is impaired by higher levels of sulfur.
8.2.3.3 Diesel Oxidation Catalysts
Diesel oxidation catalysts (DOCs) are the most common aftertreatment emissions control technology found in current diesel vehicles. DOCs are very similar to the earliest catalysts used for gasoline engines. Oxidation catalysts work by oxidizing CO, HC and the soluble organic fraction of the PM to CO2 and H2O in the oxygen rich exhaust stream of the diesel engine.
When sulfur is present in the fuel, DOCs also increase the oxidation rate of SO2, leading to increases in sulfate nanoparticle emissions. Sulfate conversion depends on overall catalyst efficiency, with more efficient catalysts capable of converting nearly 100% of the SO2 in the exhaust to sulfate. Generally, one should restrict the use of DOCs to areas which have fuel sulfur levels of 500 PPM or below. With low sulfur fuel, a DOC can reduce PM emissions by 25 to 30%.
8.2.3.4 Diesel Particulate Filters
Diesel particulate filters (DPFs) already reliably demonstrate over 95% efficiency with near-zero sulfur fuel use. They are also capable of reducing the total number of particles emitted to levels similar or even slightly lower than those of gasoline engines. One important area of research—the area most impacted by sulfur levels—is the passive regeneration or cleaning of the collected particles from the filter surface. Filters need to be cleaned, ideally without human intervention, before reaching capacity in order to maintain vehicle performance and fuel and filter efficiency.
The Continuously Regenerating Diesel Particulate Filter (CR-DPF) and the Catalyzed Diesel Particulate Filter (CDPF) are two examples of PM control with passive regeneration. The CR-DPF and CDPF devices were found to achieve 95% efficiency for control of PM emissions with 3 ppm sulfur fuel (see DECSE study). But efficiency dropped to zero with 150 ppm sulfur fuel and PM emissions more than double over the baseline with 350 ppm sulfur fuel. The increase in PM mass comes mostly from water bound to sulfuric acid. Soot emissions also increase with higher sulfur fuel but even with the 350 ppm sulfur fuel DPFs maintain around 50% efficiency for non-sulfate PM. The systems eventually recover to original PM control efficiency with return to use of near-zero sulfur fuels, but recovery takes time due to sulfate storage on the catalyst.
As noted by TERI in its recent study of PM filters and low sulfur fuel in Mumbai, “CRT technology proved to be highly effective in reducing PM emissions from ULSD powered BS -II buses. It is, however, important to highlight that CRT is very sensitive to the sulphur content in diesel. According to JM, its manufacturer, a CRT can work effectively only if it is used in a modern diesel bus running on not more than 50 ppm sulphur–diesel or ULSD. The conversion efficiency of the CRT, after it was stabilized, was found to be 95% for SOF and over 98% for IOF . . . The CRT was very effective in reducing the free acceleration smoke too” (“Workstream 1,” 2004).
Sulfur also increases the required temperature for regeneration of the filter. In moving from 3 to 30 ppm sulfur fuel, the exhaust temperatures required for regeneration increase by roughly 25°C. The CDPF requires consistently higher temperatures but holds stable above 30 ppm, while the CR-DPF requires ever-increasing temperatures.
Work continues to develop filters that are less sensitive to sulfur in fuels. One emerging technology, the so-called flow through filter that achieves about 50% PM reduction is, for example, less sensitive to sulfur than the wall flow filter which can achieve 90% or greater PM reductions.
8.2.3.5 NOx Control Systems
Many diesel engines rely on injection timing retard to meet the NOx standards currently in place. Injection timing retard reduces the peak temperature and pressure of combustion, thus reducing NOx formation. Unfortunately, this solution both increases PM emissions and significantly decreases fuel economy. For example, NOx emissions can be decreased by 45% by retarding the injection timing 8 degrees, but this results in a 7% loss in fuel economy. Injection timing retard is not impacted by sulfur in fuel.
Exhaust gas recirculation (EGR), another NOx control strategy which is being used extensively today, is only indirectly impacted by fuel sulfur. Two very different technologies—NOx adsorbers and selective catalytic reduction (SCR) systems—are the most likely alternatives for stringent NOx control.
8.2.3.6 Exhaust Gas Recirculation
Major advances in diesel NOx control have been made with exhaust gas recirculation (EGR), which lowers combustion temperatures and thus reduces thermal NOx formation. Fuel sulfur does not impact emissions from EGR systems in diesel engines, but it does hinder system durability and reliability due to sulfuric acid formation. In order for EGR to be effective, the exhaust gases must be cooled, which causes sulfuric acid to condense in the recirculation system. Acid formation raises system costs, due to the need for premium components and increased maintenance costs.
8.2.3.7 Selective Catalytic Reduction (SCR)
SCR is emerging as the leading NOx reduction technology in Europe to meet Euro IV and Euro V heavy-duty diesel standards. SCR uses a reducing agent, injected into the exhaust gas before the catalyst, to achieve high rates of NOx conversion in the oxygen-rich exhaust. SCR systems are completely ineffective if the urea reagent is not added and thus requires great attention to in use enforcement and monitoring when this technology is used. European regulators are taking steps to require fail-safe systems that will significantly degrade vehicle performance if the urea tank is not filled. Stationary systems have over 90% conversion efficiency and are widely used for diesel generators and power production.
Sulfur does not reduce conversion efficiency in SCR systems as directly as in other advanced control technologies, but emissions are impacted in a couple of ways. Fuel sulfur will increase the PM emissions from the downstream oxidation catalyst. Sulfur reactions in urea-based SCR systems can also form ammonium bi-sulfate, a severe respiratory irritant.
8.2.3.8 NOx Adsorbers
NOx adsorbers are also known as NOx storage catalysts or lean NOx traps. NOx adsorber systems are still under development but are expected to be introduced in the US for some engines in 2007. They have demonstrated 95% efficiency in conversion of NOx to N2, with a nominal fuel penalty of 1.5%. However, without significant technological breakthroughs, it is generally recognized that this system can only operate with near zero sulfur fuels.
8.2.3.9 PM Retrofits
A growing body of data continues to show that the combination of very low sulfur fuel (usually with 50 ppm sulfur or less) and particulate filters can bring about approximately 90% reductions in PM and further substantial reductions in CO and HC from existing diesel vehicles, even after 400,000 miles of operation (“The Success of Diesel Retrofits: A Fuel Supplier Perspective”, Bob Schaefer, BP Global Fuels Technology, December 2003). To obtain these reductions however requires a careful matching of the technology to the vehicles with special attention given to operational patterns and exhaust temperature profiles.
Diesel oxidation catalysts can also be retrofitted to existing diesel vehicles as is occurring in Hong Kong, with overall PM reduction on the order of 25%.